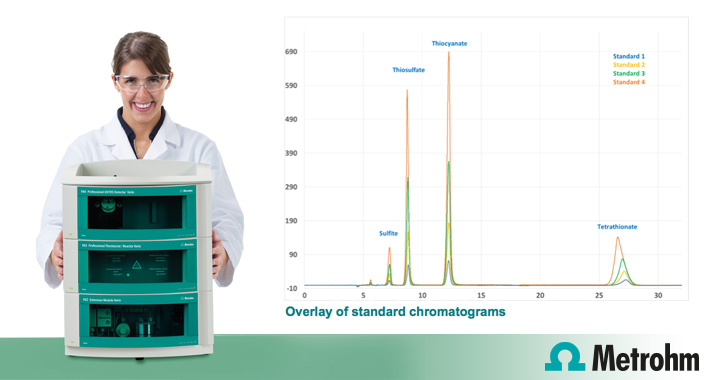
In exception of sulphate, most of the sulphur species can be determined by direct UV detection at about 215 nM.
Health and safety pressures is motivating the gold mining industry to develop a substitute to cyanide for leaching processes. The drive is mainly based on the high toxicity of cyanide.
Sulphur speciation by Ion Chromatography
The chemistry involved in the thiosulphate leaching process is more complex and less robust than the cyanide leaching process, and is therefore more difficult to optimise and more sensitive to operate. The leach process involves a chemical reaction between metallic gold and the thiosulphate anion, with oxygen as the oxidant, ammonium and copper ions as unconsumed catalysts in the reaction. Thiosulphate forms strong complexes with Au(I), Cu(I), Cd(II), Bi(II), Hg(II) and Fe(III). In the presence of ammonium thiosulphate with oxygen, gold complexes with thiosulphate as follows:
4Au + 8S2O32– + O2 + 2H2O → 4 Au(S2O3)2 3– + 4OH–
While it’s relatively easy, to establish the amenability of an ore to cyanide leaching and optimise the process, the thiosulphate chemistry presents technical challenges and requires careful optimisation. Thiosulphate leaching is a sensitive process that requires both dependent and independent optimisation of each of the chemical components of the leach reaction in order to maximise gold recovery and minimise reagent losses.
This MEP application provided below aims is to determine all the common sulphur species, in exception of sulphate via ion exchange chromatography, using perchlorate as a mobile phase and utilising direct UV absorption as detection technique.
Perchlorate was chosen as the main constituent in the mobile phase, given the fact that the samples might contain relative high concentrations of transition and heavy metals.
Most of the metal perchlorates are highly soluble in water. Hence why this mobile phase was chosen in order to diminish potential precipitation of metals on the anion exchange column.