Fully automatic determination of the total, calcium and magnesium hardness of water samples using photometric titration.
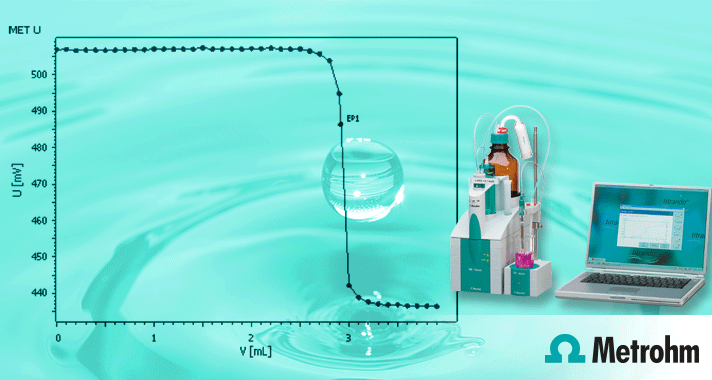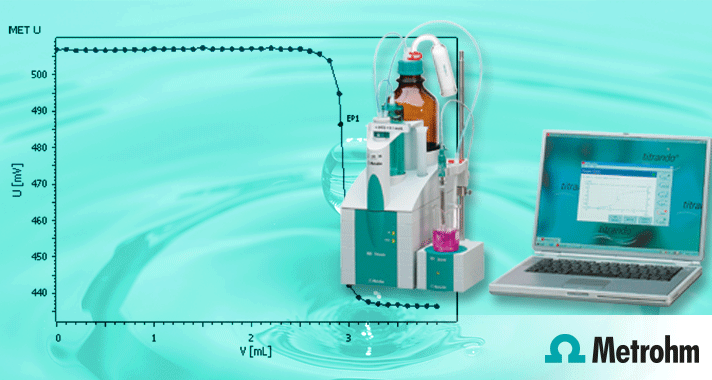
The total hardness is the sum of calcium, magnesium, barium and strontium ions. Since in drinking water only calcium and magnesium are present in significant amounts, the hardness is entirely determined by their content. The total hardness can be determined by potentiometric titration with ion-selective electrodes.
Water hardness by photometric titration
Metrohm is pleased to present the new titration application for fully automatic determination of the total, calcium and magnesium hardness of water samples by using photometric titration method.
This Application Note describes the determination of overall hardness in addition to that of calcium and magnesium in water samples by means of the Optrode (610 nm). The total hardness is determined with EDTA as titrant and Eriochrome Black T as indicator. The calcium hardness is in turn determined with EDTA and with calconcarboxylic acid indicator. The magnesium hardness is the difference between total hardness and calcium hardness. The application note is available for free download below.
Now with the variable wavelength Optrode from Metrohm, calcium and magnesium titrations are made simple. With a Metrohm Titrator such as the 916 Ti Touch or a Titrando, in combination with the Optrode, this titration can be easily automated. No longer requiring the operator to visually see the detection of the equivalence point.